
The FDA doesn't just approve generic drugs and move on. It runs a complex, data-driven system to monitor safety after approval, catching manufacturing flaws and rare side effects before they become widespread. Here's how it works - and where it still falls short.

Learn how to use pharmacy apps to spot dangerous drug interactions before they happen. Compare top tools, avoid common mistakes, and keep your meds safe with expert-backed tips.

How federal and state laws shape generic drug payments through MAC pricing, PBM spread, Medicare Part D tiers, and the new $2 Drug List. Understand why your copay changes - and how to pay less.

If you get the wrong medication from the pharmacy, stop taking it immediately and call your doctor. Preserve all evidence, report the error, and know your legal rights to protect yourself and prevent future mistakes.

Pharmacists play a vital role in spotting and reporting generic drug problems like therapeutic inequivalence and manufacturing issues. Learn what to report, how to do it, and why your reports matter to patient safety.

Learn the exact temperature and time conditions required for pharmaceutical stability testing under ICH Q1A(R2) guidelines. Understand long-term, accelerated, and refrigerated protocols used globally to ensure drug safety and efficacy.

Learn how to properly document safety alerts on medication lists to prevent serious errors with high-risk drugs like insulin, opioids, and anticoagulants. Follow proven steps from ISMP guidelines.
U.S. generic drug prices are 33% lower than in other wealthy countries, but brand-name drugs cost up to five times more. Here's how global pricing works - and what it means for your prescriptions.

Bioequivalence studies prove generic drugs work the same as brand-name versions by measuring how quickly and how much of the drug enters the bloodstream. This step-by-step process ensures safety, effectiveness, and cost savings.
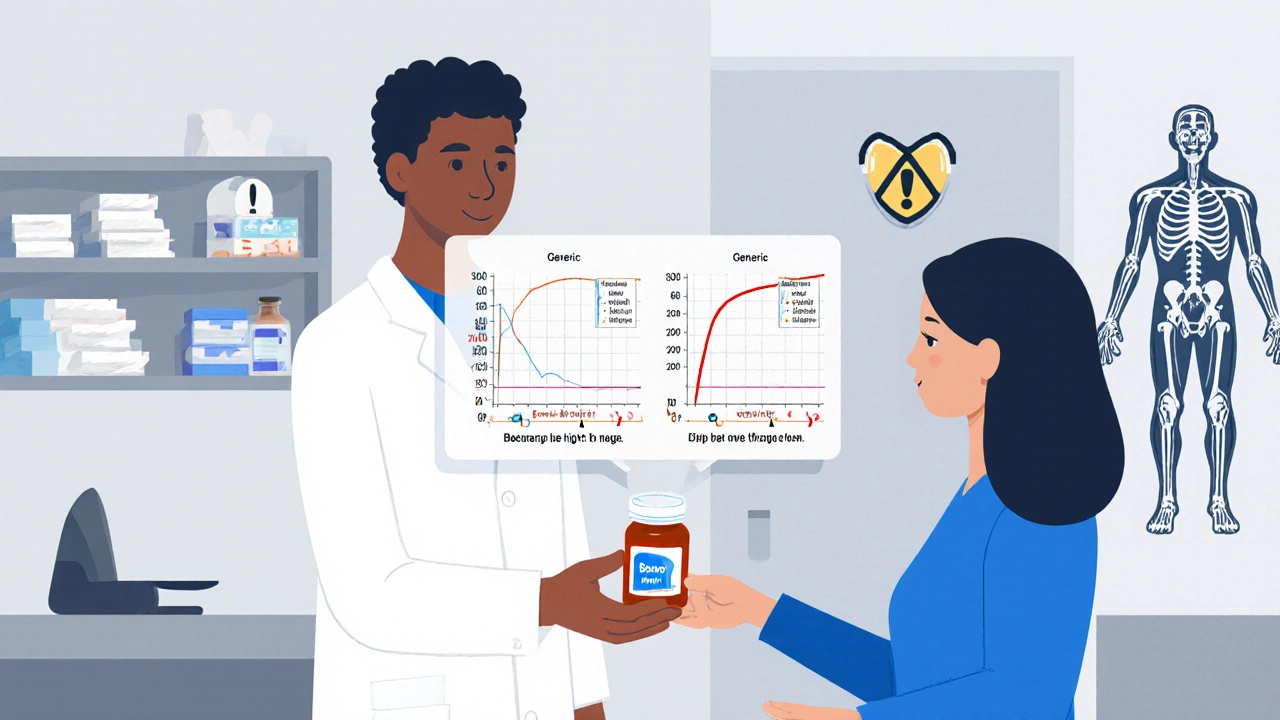
Therapeutic Drug Monitoring is critical for patients on generic narrow therapeutic index (NTI) drugs, where small changes in blood levels can cause toxicity or treatment failure. Learn when and why TDM saves lives.