Imagine a patient sitting in your exam room, staring at a prescription for a generic medication with deep skepticism. They've heard a rumor that the "off-brand" version isn't as strong or is made in a less regulated facility. If the clinician in the room hesitates or shares that doubt, the patient's trust in the treatment evaporates. This is the "nocebo effect" in action, and it's a primary reason why some patients simply never start their medication. The problem isn't just patient misconception; it's a gap in provider confidence.
While generic drugs make up about 90% of prescriptions dispensed in the U.S., a surprising number of healthcare professionals still harbor doubts about their equivalence. When a doctor isn't 100% sure how a generic is approved, they can't effectively advocate for it. Improving provider education on generics isn't just about sharing pamphlets; it's about dismantling long-held myths and replacing them with hard data to improve patient adherence and lower healthcare costs.
| Metric | Impact of Educated Prescribing |
|---|---|
| Patient Adherence | 3.2x higher when clinicians explicitly endorse equivalence |
| Therapy Initiation | Patients are 35% more likely to start therapy with generics |
| Side Effect Reports | 18% fewer patient-reported side effects (reduced nocebo effect) |
| Prescribing Rates | 29% higher generic initiation for new prescriptions |
Closing the Knowledge Gap: What Clinicians Actually Misunderstand
It's a common assumption that every doctor knows exactly how generics work. In reality, there are significant blind spots. For example, many prescribers mistakenly believe that generic drugs must have the exact same inactive ingredients as the brand name. In truth, Inactive Ingredients is the list of excipients-like binders or fillers-that can differ between generic and brand versions without affecting the drug's safety or efficacy. Around 45% of prescribers get this wrong, leading to unnecessary concerns about allergies or fillers.
Another hurdle is the misunderstanding of Bioequivalence, which is the requirement that a generic drug delivers the same amount of active ingredient into the bloodstream at the same rate as the brand-name drug. Some clinicians believe the FDA allows for a 20-25% difference in active ingredients. This is a myth. The standards are much tighter, ensuring that the drug performs the same way in the body regardless of the label.
Then there's the "brand-name habit." Some attending physicians still use brand names exclusively, which creates a dangerous environment for medical residents. When a resident learns a drug by its generic name but an attending calls it by a brand name, it can lead to prescribing errors-like accidentally ordering two different versions of the same medication.
The Science of Approval: Understanding the FDA Standards
To feel confident in a generic, providers need to understand the Abbreviated New Drug Application (ANDA). This is the process used by generic manufacturers to gain FDA approval without repeating the massive clinical trials the original brand-name drug went through. Instead, they prove the drug is therapeutically equivalent.
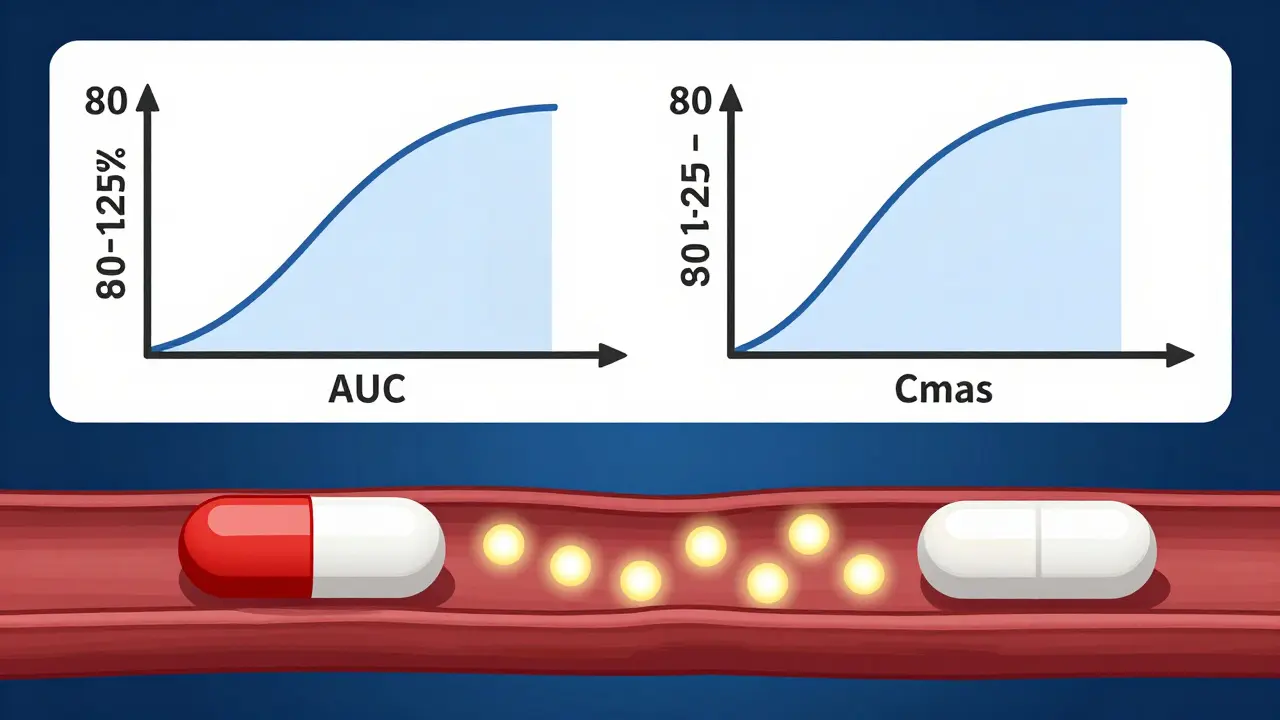
The gold standard for this proof is the 90% confidence interval rule. For a drug to be approved, the ratio of the population geometric means for the Area Under the Curve (AUC) and the maximum concentration (Cmax) must fall between 80% and 125%. In plain English: the drug's absorption and peak concentration must be nearly identical to the original.
Clinicians can verify this using the Orange Book, officially known as the Approved Drug Products with Therapeutic Equivalence Evaluations. The Orange Book is the FDA's primary resource for identifying which generic versions are therapeutically equivalent to a reference listed drug. If a drug has an "A" rating, it is considered equivalent and can be substituted. A "B" rating means it lacks that equivalence.
Effective Strategies for Provider Training
Traditional methods like handing out static fact sheets are largely ineffective. Research shows a massive difference in how we learn: clinicians who engage in interactive, case-based education see 42% higher knowledge retention over six months compared to those who just read a flyer. The key is moving from passive information to active problem-solving.
Spaced repetition is also critical. A single eight-hour seminar is quickly forgotten. Instead, breaking the education into four 90-minute sessions over half a year resulted in 52% higher retention. This allows the knowledge to move from short-term memory to long-term clinical practice.
Modern systems are now integrating this education directly into the workflow. Rather than requiring a doctor to leave their office to attend a webinar, Electronic Health Records (EHR) are being used to trigger real-time prompts. When a doctor selects a brand-name drug, the system can provide a quick pop-up confirming the generic's bioequivalence, acting as a "just-in-time" educational tool.
The High-Stakes Areas: Psychiatric and Chronic Care
Some medical specialties see more benefit from this education than others. In psychiatry, the nocebo effect is particularly potent. If a patient believes a generic antidepressant is inferior, they may report more side effects or perceive the drug as ineffective, leading to early discontinuation. When a psychiatrist explicitly endorses the generic's equivalence, the patient is far more likely to stay on the medication.
Chronic disease management, such as diabetes or hypertension, is another critical area. Here, the barrier isn't just trust-it's cost. Patients are 35% more likely to start therapy when they know a cheaper generic option is available. Providers who are educated on the economic benefits of generics can help their patients avoid "cost-related non-adherence," where a patient skips doses because they can't afford the brand name.
However, the education must be nuanced. There is a difference between small-molecule generics and Biosimilars. While a generic is a chemical copy, a biosimilar is a highly similar version of a complex biologic drug. Only about 31% of providers can correctly explain this difference, highlighting a new frontier for clinician education in oncology and rheumatology.

Overcoming Common Barriers to Implementation
The biggest obstacle to improving clinician knowledge is, unsurprisingly, time. Nearly 89% of physicians cite time constraints as the reason they don't discuss generic options in depth with patients. To solve this, education must be integrated, not added. This means incorporating generic drug education into residency programs so that the next generation of doctors doesn't have to "unlearn" brand-name habits.
Another barrier is the lack of awareness of existing resources. Many providers aren't aware of the FDA's Generic Drugs Stakeholder Toolkit or the specific handouts designed for prescribers. Moving these resources from a hidden website to a shared clinical dashboard can bridge this gap.
| Format | Retention Rate | Clinical Impact | Best For... |
|---|---|---|---|
| Static Fact Sheets | Low | Minimal | Quick reference |
| Case-Based Learning | High (+42%) | Significant | Medical residents |
| EHR Integrated Alerts | Moderate | High (24% uptake) | Daily practice |
| VR Simulation | High | Confidence Boost | Patient communication |
Next Steps for Health Systems and Clinicians
For health systems, the goal should be a mandatory onboarding module for new prescribers that focuses on the 80-125% bioequivalence rule and the use of the Orange Book. Integrating generic prescribing rates as a quality metric (as seen in the 2025 MIPS goals) provides the necessary incentive for doctors to prioritize this knowledge.
For individual clinicians, the most immediate action is to change the language used with patients. Instead of saying "You can take the generic if you want," try "I'm prescribing the generic version because it is therapeutically identical to the brand name and will work exactly the same way." This simple shift in endorsement is the strongest predictor of whether a patient will actually take their medicine.
What is the difference between a generic and a biosimilar?
Generics are chemically identical copies of small-molecule drugs. Biosimilars are highly similar, but not identical, copies of large, complex proteins (biologics) made in living cells. While generics have the same active ingredient and structure, biosimilars have slight variations but no clinically meaningful differences in safety or potency.
How does the FDA ensure a generic is as good as the brand name?
The FDA requires generic manufacturers to prove bioequivalence. This means the drug must deliver the same amount of active ingredient to the bloodstream at a similar rate. They use a strict 80-125% confidence interval for the AUC (area under the curve) and Cmax (peak concentration) to ensure the therapeutic effect is identical.
Can inactive ingredients in generics affect the patient?
In the vast majority of cases, no. Inactive ingredients (excipients) can differ between the brand and generic versions. However, they must be safe and cannot interfere with the drug's absorption or effectiveness. A small number of patients may have a specific allergy to a particular filler, but this is rare and not a reflection of the drug's overall efficacy.
What is the Orange Book and how should providers use it?
The Orange Book is the FDA's official guide to therapeutic equivalence. Providers can look up a brand-name drug to see which generics are rated "A" (equivalent) or "B" (not equivalent). If a drug is "A" rated, the pharmacist can generally substitute it without worrying about a change in clinical outcome.
Why does provider endorsement matter more than the cost?
While cost is a driver, psychological trust is the primary barrier. When a provider expresses doubt, it triggers a nocebo effect where the patient focuses on potential failures. Direct endorsement of equivalence can make a patient 3.2 times more likely to adhere to their treatment plan compared to a passive recommendation.

