Pharmacogenomic Risk Calculator
This tool estimates your potential risk of adverse drug reactions based on common genetic variants affecting medication metabolism. Results are for educational purposes only and should not replace professional medical advice.
Select Your Medication
Select Your Genetic Status
Have you ever taken a medication that didn’t work-or made you feel worse? You’re not alone. For many people, the same pill that helps one person do nothing for another, or even cause serious side effects. The reason? Your genes. Pharmacogenomics is the science that explains why your DNA matters when it comes to drugs. It’s not science fiction. It’s happening right now in hospitals, clinics, and even in your own medicine cabinet.
Why Your Body Processes Drugs Differently
Everyone’s body handles drugs differently. One person takes an antidepressant and feels better in weeks. Another takes the same pill for months and feels nothing-or gets dizzy, nauseous, or worse. Why? Because of how your body breaks down and responds to medication. This is where pharmacogenomics comes in. It looks at your genes to figure out how fast or slow your liver processes drugs, how your brain reacts to them, and whether you’re at risk for dangerous side effects.
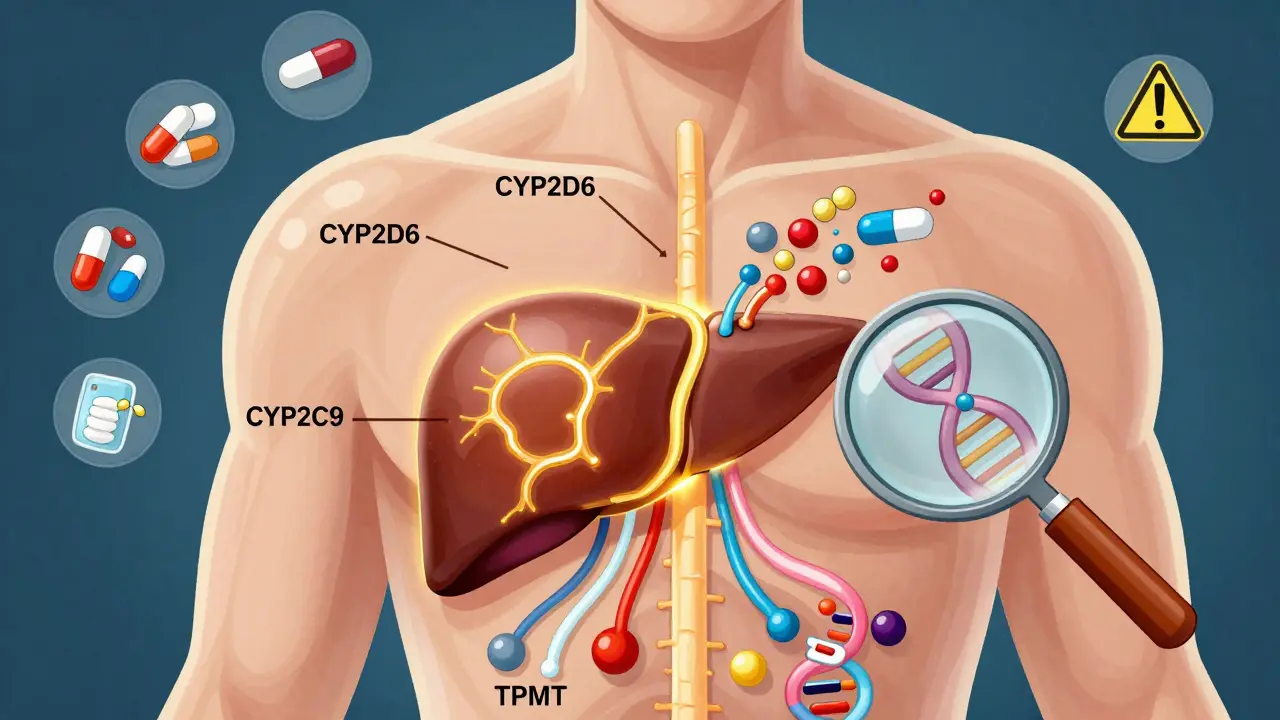
Most of this comes down to enzymes-special proteins that act like molecular scissors, cutting drugs apart so your body can get rid of them. The most important family of these enzymes? The cytochrome P450 system. Of these, CYP2D6 is the superstar. It handles about 25% of all prescription drugs, including common antidepressants like fluoxetine (Prozac), painkillers like codeine, and beta-blockers like metoprolol. But here’s the catch: people have different versions of the CYP2D6 gene. Some have extra copies (ultra-rapid metabolizers), some have broken copies (poor metabolizers), and most have normal function.
If you’re an ultra-rapid metabolizer, codeine turns into morphine too fast. That can lead to dangerous breathing problems-even if you take the right dose. If you’re a poor metabolizer, the drug just sits in your system, building up to toxic levels. That’s why some people get sick on standard doses while others need double the amount to feel anything.
The Real-World Impact: Less Trial and Error
Before pharmacogenomics, doctors guessed. They’d start you on one antidepressant, wait to see if it worked, then switch if it didn’t. On average, patients try three or four different drugs before finding one that helps. That can take months. During that time, symptoms get worse. Some people even try to end their lives.
Now, imagine knowing your genetic profile before the first prescription. A 2022 study in JAMA looked at 1,838 patients with depression. Those who got genetic testing had a 26.9% higher chance of going into remission compared to those who didn’t. Side effects dropped by nearly 30%. That’s not a small improvement-it’s life-changing.
It’s not just mental health. Warfarin, a blood thinner used after heart attacks or strokes, has a narrow safety window. Too little, and you risk a clot. Too much, and you bleed internally. For decades, doctors used a one-size-fits-all starting dose. Now, testing for CYP2C9 and VKORC1 genes cuts the time to reach the right dose by over two days and reduces dangerous bleeding by 31% in the first month.
Genes That Save Lives
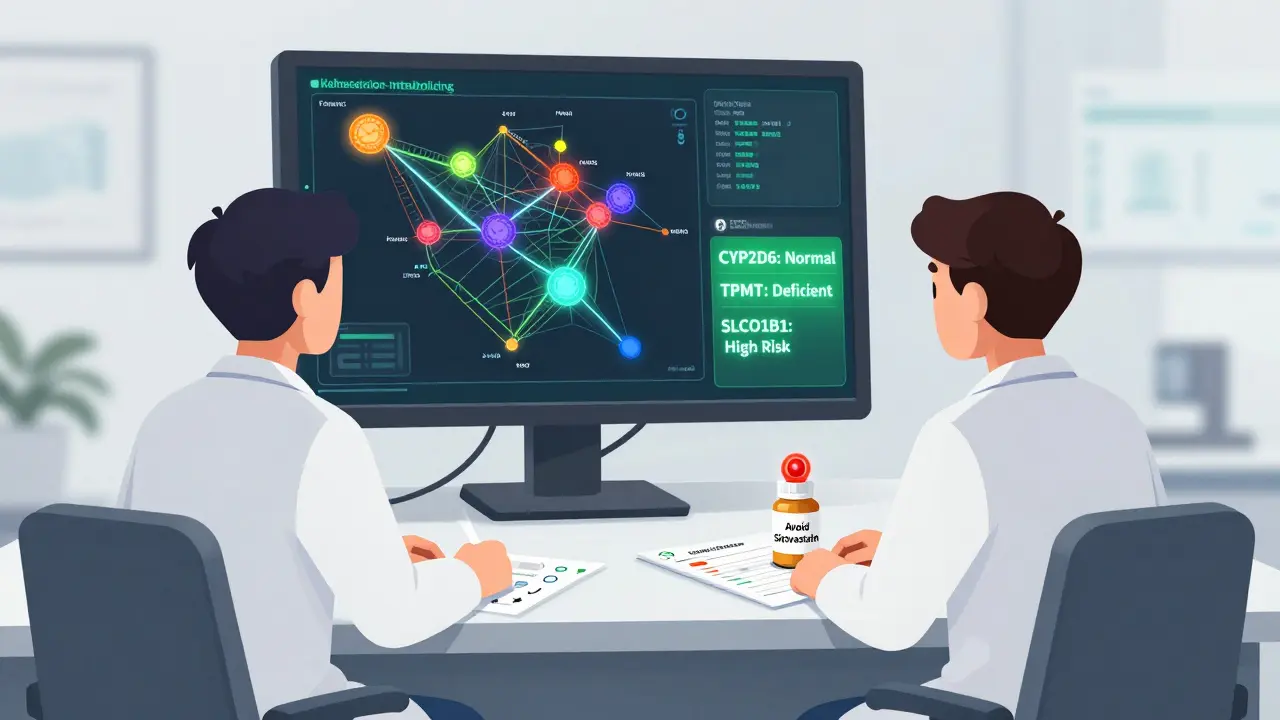
Some gene-drug pairs are so critical that testing is now standard practice. Take TPMT. This enzyme breaks down thiopurine drugs used in leukemia and autoimmune diseases. About 0.3% of people have a mutation that makes them unable to process these drugs. Without testing, they get hit with full doses-and their bone marrow shuts down. The result? Life-threatening infections, hospitalization, even death. A simple genetic test before treatment prevents this entirely.
Another example is DPYD. This gene affects 5-fluorouracil (5-FU), a chemotherapy drug used for colon, breast, and other cancers. About 0.2% of patients have a variant that causes severe toxicity-vomiting, diarrhea, nerve damage, and death. Testing for DPYD before chemo has prevented hundreds of deaths in the U.S. alone. The FDA now recommends this test before starting treatment.
Even statins, the cholesterol-lowering drugs millions take daily, have a genetic risk. A variant in SLCO1B1 increases the chance of muscle pain and damage from simvastatin by 4.5 times. For people with this variant, doctors can switch to a safer statin before any muscle damage occurs.
Why It’s Not Everywhere Yet
So why don’t we all get tested? The answer is complicated. First, cost. A full pharmacogenomic panel runs $250-$500. While some insurance plans cover it-especially for high-risk drugs like clopidogrel or thiopurines-many don’t. Patients report waiting 14+ days just to get approval. Some tests get denied outright.
Second, doctors aren’t trained for it. A 2023 survey found that 68% of clinicians feel more confident prescribing after seeing genetic results-but nearly half say their electronic health records don’t support it. No alerts. No easy way to see the results. No guidance on what to do next. That’s like giving someone a map but no compass.
Third, the data is skewed. Over 90% of pharmacogenomic studies have been done on people of European descent. That means the guidelines we use today may not work for Black, Asian, Indigenous, or Hispanic patients. A 2023 study in Nature Genetics found that a common CYP2D6 variant linked to poor metabolism in Europeans is rare in African populations-and vice versa. Without diverse data, we risk misjudging risk for millions.
What’s Being Done
Change is coming. The Mayo Clinic’s RIGHT Study tested over 10,000 patients and found a 30% drop in adverse drug reactions. They saved $1,200 per patient per year. The VA has tested over 100,000 veterans and cut hospitalizations by 22%. Vanderbilt’s PREDICT program has been screening patients since 2012 and saved $1.9 million in avoided ER visits.
The FDA approved its first next-generation PGx test in January 2023. OneOme’s RightMed Comprehensive checks 27 genes and 350+ medications. The NIH just launched a $190 million project to expand testing in underrepresented groups. And the World Health Organization added pharmacogenomics to its list of essential diagnostics in 2023.
More insurers are covering it too. In 2020, only 28% of commercial plans covered any PGx test. By 2023, that jumped to 65%. Medicare Advantage plans now cover 87% of them.
What You Can Do Now
You don’t need to wait for your doctor to order a test. If you’ve had a DNA test from 23andMe or Ancestry, you can download your raw data and use services like GeneSight or Pillcheck to analyze your PGx profile. They’ll tell you how your genes affect medications you’re already taking-or might take in the future.
Or, if you’re starting a new drug-especially an antidepressant, painkiller, blood thinner, or chemo agent-ask your doctor: “Has my genetic profile been considered?” If they’re unsure, ask for a referral to a clinical pharmacist or genetic counselor. They’re trained to interpret these results.
And if you’ve had bad reactions to medications in the past, keep a record. Note the drug, the dose, and what happened. That history is gold for a pharmacist or genetic specialist.
The Future Is Personal
Pharmacogenomics isn’t about replacing your doctor. It’s about giving them better tools. In the next five years, it’s likely that getting your PGx profile will be as routine as getting your blood type. Imagine walking into a clinic at age 18, getting a simple cheek swab, and having your entire drug response profile stored in your medical record. Every prescription you get from then on is tailored to you.
That future is closer than you think. And for people who’ve spent years trying drug after drug with no luck, it’s not just science-it’s hope.
What is pharmacogenomics?
Pharmacogenomics is the study of how your genes affect how your body responds to medications. It combines genetics and pharmacology to predict whether a drug will work for you, how much you need, and if you’re at risk for side effects. It’s not about diagnosing disease-it’s about matching the right drug to the right person.
Which genes are most important for drug metabolism?
The most important genes are those that code for drug-metabolizing enzymes. CYP2D6 handles about 25% of all prescription drugs, including antidepressants and opioids. CYP2C19 affects clopidogrel (Plavix) and some antidepressants. CYP2C9 and VKORC1 are key for warfarin dosing. TPMT and DPYD are critical for chemotherapy drugs. SLCO1B1 affects statin safety. These six genes account for the majority of clinically actionable drug-gene interactions.
Is pharmacogenomic testing covered by insurance?
Coverage is growing but still inconsistent. As of 2023, 87% of Medicare Advantage plans and 65% of private insurers cover at least one PGx test-usually for high-risk drugs like clopidogrel, thiopurines, or warfarin. But coverage for broader panels or pre-emptive testing is rare. Many patients still face denials or long delays. Always check with your insurer before testing.
Can I get tested without a doctor’s order?
Yes. Companies like 23andMe and Ancestry offer limited pharmacogenomic reports as part of their health services. You can also upload your raw DNA data to third-party services like GeneSight, Pillcheck, or MyGeneRank. These services analyze your genes for drug responses and provide reports you can share with your doctor. However, they don’t replace clinical testing and may not be as comprehensive.
Why doesn’t everyone get tested if it works so well?
Three big reasons: cost, integration, and bias. Testing costs $250-$500, and insurance doesn’t always pay. Electronic health records don’t always show results clearly, making it hard for doctors to use them. And most research has been done on white populations, so guidelines may not apply to others. Until these issues are fixed, testing won’t be routine-even though it saves lives.
What should I do if I’ve had bad reactions to medications before?
Write it down. Note the drug name, dose, and what happened-rash, nausea, dizziness, hospitalization. Bring that list to your doctor or pharmacist. Ask if pharmacogenomic testing could help explain it. Many adverse reactions have genetic roots. Knowing your profile can prevent future harm and help you avoid drugs that won’t work for you.

becca roberts
March 19, 2026 AT 22:58So let me get this straight-we’ve known for years that people metabolize drugs differently, but we’re only now starting to test for it? And we’re acting like this is some revolutionary breakthrough? I’ve had my DNA done through 23andMe. My CYP2D6 is *wildly* ultra-rapid. Took me three tries to find an antidepressant that didn’t make me feel like I’d been hit by a truck. Meanwhile, my cousin takes the same pill and says it’s ‘life-changing.’ Yeah, no shit. It’s not magic. It’s biology. And we’re still treating it like a luxury upgrade instead of basic healthcare.
Andrew Muchmore
March 21, 2026 AT 12:59Testing works. It saves money. It saves lives. Stop overcomplicating it. If you’re on a drug with known gene interactions, get tested. No debate. No waiting. Just do it.
Paul Ratliff
March 23, 2026 AT 09:48bro i got my 23andme results last year and it told me i’m a poor metabolizer for codeine. so i stopped taking it. no more dizziness. no more nausea. just… peace. i told my doc and he was like ‘huh, interesting’ and didn’t change anything. like bro, you have the data. use it. i’m not asking for a miracle. just don’t poison me with your guesswork.
Kathy Underhill
March 24, 2026 AT 10:23There’s a quiet revolution happening in clinical pharmacology. We’re moving from population-based dosing to individualized therapy. The science is solid. The data is overwhelming. What’s missing isn’t evidence-it’s infrastructure. Health systems need to integrate genetic data into EHRs with actionable alerts, not buried in PDFs. And clinicians need training-not just on the genes, but on how to communicate risk without causing panic. This isn’t sci-fi. It’s the next logical step in medicine.
Gaurav Kumar
March 25, 2026 AT 14:35India has been doing personalized medicine for decades-Ayurveda, herbal combinations, pulse diagnosis. Why are Westerners only now waking up to the idea that one-size-fits-all doesn’t work? Your DNA isn’t a Western invention. Your arrogance is. We’ve known this for centuries. You just didn’t bother to study it because it wasn’t profitable enough. Now that Big Pharma sees dollar signs, suddenly it’s ‘groundbreaking.’
Sanjana Rajan
March 26, 2026 AT 08:46Who’s funding this? Big Pharma? Let me guess-next they’ll sell us ‘DNA-optimized’ pills for $200 a dose. And don’t get me started on the data. They’re using white people’s genes as the default. What about us? My aunt died on warfarin because her ‘high-risk’ variant was never studied in South Asians. They didn’t even test her. They just said ‘you’re fine.’ Fine? FINE? You call that medicine? This isn’t science-it’s a scam with a lab coat.
Nicole Blain
March 27, 2026 AT 23:33me: "i had a bad reaction to prozac"
doctor: "oh, weird, it works for everyone"
me: *pulls up 23andme report*
doctor: "...huh. we’ll try something else." just... please stop pretending this isn’t a thing. it’s not magic. it’s just science that finally caught up to reality. 🤷♀️💊
Prathamesh Ghodke
March 29, 2026 AT 12:59My mom took three different SSRIs before they finally tested her CYP2C19. Turned out she was a poor metabolizer. Switched to sertraline. Two weeks later, she was laughing again. I cried. Not because it was expensive. Not because it was new. Because we almost lost her to a system that refused to see her as anything but a statistic. This isn’t about tech. It’s about seeing people. Not numbers. Not races. Just… humans.
SNEHA GUPTA
March 31, 2026 AT 02:32The real tragedy isn’t that we don’t test-it’s that we’ve normalized suffering. We accept that people must endure months of depression, nausea, dizziness, or worse before someone considers their biology. We call it ‘trial and error.’ I call it medical negligence. Pharmacogenomics isn’t an option. It’s a moral imperative. And if we wait for insurance to catch up, we’ll be burying more people before we fix this.
Paul Ratliff
April 1, 2026 AT 14:21^^^ this. my doc didn’t even know what CYP2D6 stood for. i had to google it and show him. he was like ‘oh, so i’m supposed to know this now?’ yeah. you are. you’re the one holding the pen. don’t make me your lab rat.