When a brand-name drug loses its patent, it doesn’t just disappear from shelves. Instead, something unexpected happens: the same company that made the original drug starts selling an almost identical version - but without the brand name. This isn’t a mistake. It’s a calculated move called an authorized generic. And it’s changing how we think about generic drugs, pricing, and competition in the U.S. drug market.
What Exactly Is an Authorized Generic?
An authorized generic is the exact same medication as the brand-name drug - same active ingredients, same inactive ingredients, same pills, same manufacturing process. The only difference? It’s sold under a different label, often with no brand name at all. It’s not a copy. It’s the real thing, just repackaged.For example, if you’ve ever taken Concerta for ADHD, you might have seen a pill that looked identical but was labeled as "methylphenidate ER" instead. That’s an authorized generic. It came from the same factory, on the same production line, as the original. The FDA confirms this: authorized generics are considered therapeutically equivalent because they are literally the same product.
This is different from traditional generics. Those are made by other companies that reverse-engineer the drug. They must prove they’re bioequivalent - meaning they work the same way in the body - but they can use different fillers, dyes, or coatings. Sometimes, those small differences matter. For drugs with a narrow therapeutic index - like levothyroxine or warfarin - even tiny variations can cause side effects or reduced effectiveness. Authorized generics avoid that risk entirely.
Why Do Brand Companies Do This?
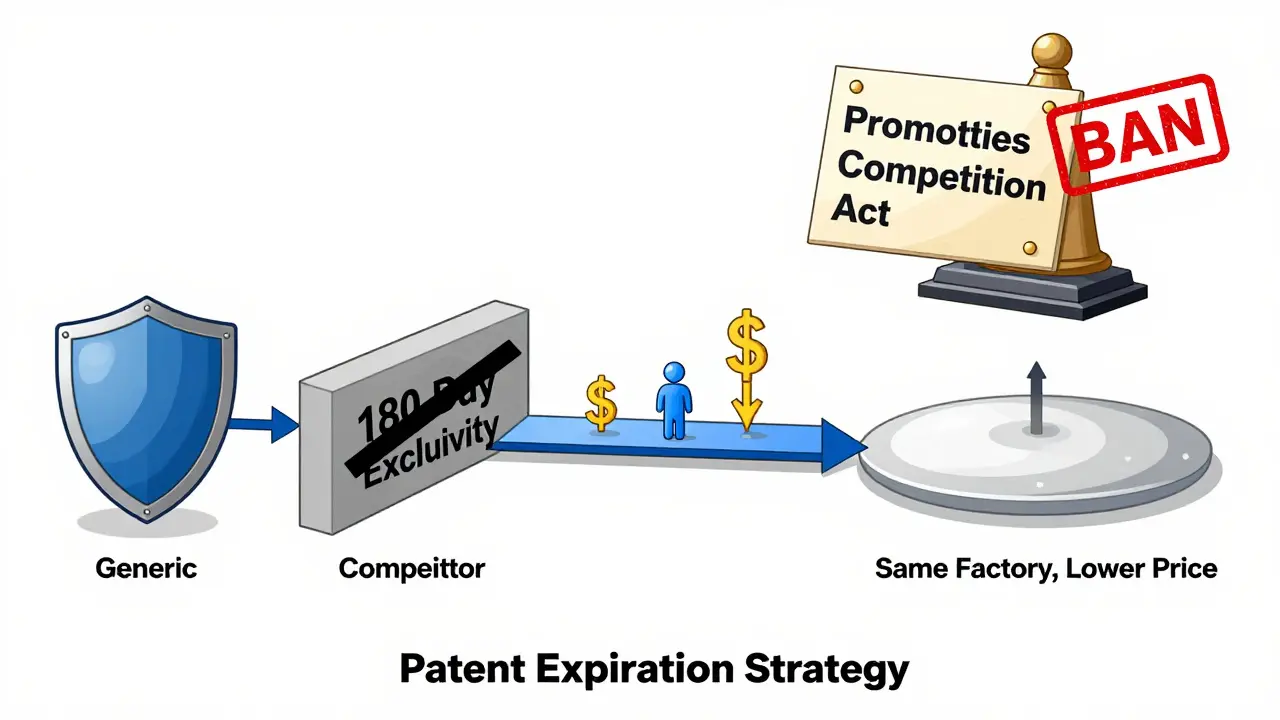
It sounds counterintuitive. Why would a company that spent millions developing a drug turn around and sell a cheaper version of it? The answer lies in timing and control.When a patent expires, the first generic company to file gets 180 days of exclusivity under the Hatch-Waxman Act. During that time, they’re the only generic on the market. Prices stay high. But if the brand company waits, it risks losing all its customers. So instead, they launch their own authorized generic - often right before or during that 180-day window.
This strategy does two things: it lowers prices for consumers and keeps the brand company in the game. According to the Federal Trade Commission’s 2011 report, when authorized generics enter the market during the exclusivity period, prices drop 15-20% faster than in markets without them. That’s a win for patients and insurers. But it also means the brand manufacturer still collects revenue - just under a different label.
Between 2010 and 2019, there were 854 authorized generic launches, peaking in 2014. By 2025, that number had grown to over 1,200. Nearly half of the top-selling brand drugs facing patent expiration between 2015 and 2020 had authorized generics launched within six months. That’s not coincidence. It’s a playbook.
How Are Authorized Generics Different From Regular Generics?
Here’s a quick breakdown:| Feature | Authorized Generic | Traditional Generic |
|---|---|---|
| Manufacturer | The original brand company | A different company |
| Active Ingredients | Identical | Identical |
| Inactive Ingredients | Identical | May differ |
| Regulatory Path | Uses brand’s NDA; FDA notification only | Requires ANDA approval |
| Appears in FDA Orange Book? | No | Yes |
| Therapeutic Equivalence | Identical to brand | Proven bioequivalent |
Because authorized generics use the original brand’s New Drug Application (NDA), they don’t need to go through the full FDA approval process. They just need to notify the agency. That means faster market entry - sometimes within weeks of patent expiration. Traditional generics can take months or even years to get approved.
But here’s the catch: because they’re not listed in the FDA’s Orange Book, pharmacists can’t easily flag them as interchangeable. That creates confusion in pharmacies.
Confusion in the Pharmacy
Patients often don’t realize they’re getting the same drug they’ve always taken. One patient on Drugs.com wrote: "I got this ‘generic’ but it looks identical to the brand I used before - is this actually generic?"That’s not an isolated comment. A 2021 survey by the National Community Pharmacists Association found that 57% of independent pharmacies reported a sharp rise in patient questions about authorized generics after the FDA required clearer labeling. Pharmacists say they spend extra time explaining that the pill in the bottle might look like the brand, but it’s cheaper - and identical.
Even more troubling: 68% of pharmacists surveyed in 2020 said patients confused authorized generics with traditional generics. Some patients think they’re getting a lower-quality product. Others worry they’re being tricked. The truth? They’re getting the same medicine, often at a lower price.
Pharmacy systems are catching up. Epic Systems added a special flag for authorized generics in its 2021 update. Pharmacies using it saw identification errors drop by 67%. But not all systems have this feature. Many still rely on manual checks - and that’s where mistakes happen.
Who Benefits? Who Loses?
On the surface, everyone wins. Patients pay less. Insurers save money. The brand company keeps some revenue. But not everyone sees it that way.The Generic Pharmaceutical Association (GPhA) argues that authorized generics can delay real competition. If the brand company launches its own generic, it might discourage other companies from entering the market. Why bother if the original maker is already undercutting prices? That’s why some critics call it a "market manipulation" tactic.
Dr. Jerry Avorn from Harvard Medical School put it bluntly in a 2019 JAMA commentary: "Authorized generics benefit brand manufacturers more than consumers in the long term." He points out that while prices drop during the 180-day window, they often rebound once the authorized generic is gone - because no other generics ever showed up.
On the flip side, the FTC found that in markets with authorized generics, patients paid less overall. Express Scripts reported 28% higher utilization of authorized generics compared to traditional generics because pharmacists and patients trusted the identical formulation.
For drugs like colchicine, celecoxib, or levothyroxine - where even small ingredient changes can cause problems - authorized generics are a lifesaver. They offer consistency. That’s why adoption is highest in central nervous system drugs (67%) and lowest in antibiotics (22%).
The Future of Authorized Generics
The trend isn’t slowing down. Evaluate Pharma predicts that by 2027, 45% of major branded drugs will have authorized generics - up from 32% in 2022. The FDA’s 2025 update shows over 1,200 authorized generics now on the market.But pressure is building. In 2023, Congress introduced the "Promoting Competition in Pharmaceutical Markets Act," which would ban brand companies from launching authorized generics during the 180-day exclusivity window. If it passes, it could force companies to choose: either let the market open fully - or lose control entirely.
For now, authorized generics remain a powerful tool. They’re not a loophole. They’re a legal, FDA-approved way for brand companies to adapt. And for patients, they’re often the best option - especially if you’ve had bad reactions to traditional generics.
If you’re on a drug like levothyroxine or epilepsy medication, ask your pharmacist: "Is there an authorized generic?" You might be surprised to find out you’ve been paying more than necessary - for a pill that’s exactly the same as the one you’ve been taking all along.
Are authorized generics the same as brand-name drugs?
Yes. Authorized generics contain the exact same active and inactive ingredients as the brand-name drug. They’re made in the same facility, on the same production line, and are considered therapeutically equivalent by the FDA. The only difference is the label.
Why aren’t authorized generics listed in the FDA’s Orange Book?
Because they’re marketed under the original brand’s New Drug Application (NDA), not a separate Abbreviated New Drug Application (ANDA). The FDA only lists drugs that go through the ANDA process. Authorized generics are considered a continuation of the brand product, not a new generic entry.
Can authorized generics cause side effects?
The risk is extremely low. Since they’re chemically identical to the brand, side effects should be the same - not worse. In fact, for drugs with narrow therapeutic indexes (like thyroid or seizure meds), authorized generics may reduce side effects because they eliminate variability from different inactive ingredients found in traditional generics.
Do pharmacies automatically substitute authorized generics?
No. Unlike traditional generics, which are often substituted automatically under state laws, authorized generics are not flagged as interchangeable in most pharmacy systems. You need to ask your pharmacist if one is available - and whether it’s covered by your insurance.
Why do authorized generics sometimes cost more than traditional generics?
It’s rare, but it can happen. If the authorized generic is launched during the 180-day exclusivity window and no other generics are available, it may be the only option. In that case, it might cost more than a later-entry traditional generic. But overall, authorized generics are usually cheaper than the brand and often cheaper than traditional generics too.
What Should You Do?
If you’re taking a brand-name drug that’s lost its patent, don’t assume the generic you’re given is the cheapest or best option. Ask your pharmacist: "Is there an authorized generic?" Check your prescription label - if it lists the brand name but says "authorized generic" or "produced by [brand name]" - you’re getting the real thing at a discount.For patients on high-cost medications like those for thyroid disease, epilepsy, or depression, switching to an authorized generic could mean significant savings - and fewer side effects. For insurers and pharmacies, it’s a win too. And for the brand company? It’s still in the game.
The bottom line: authorized generics aren’t a trick. They’re a smarter way to keep prices down - without sacrificing quality. And if you’ve ever been confused about why your pill looks the same but costs less - now you know why.


Lauren Volpi
March 17, 2026 AT 21:07So let me get this straight - Big Pharma just makes the same damn pill, changes the label, and calls it a 'generic' to keep raking in cash? Brilliant. I'm not mad, I'm just impressed. They're not even pretending anymore. It's like they took the whole 'free market' thing and ran it through a blender with a smirk. And we're supposed to be grateful? For what? Getting ripped off slower?
Kal Lambert
March 18, 2026 AT 20:10Authorized generics are actually a win for patients on narrow-therapeutic-index drugs. No variability in fillers means fewer side effects. If you're on levothyroxine or warfarin, this isn't a trick - it's a safety net. Ask your pharmacist. It's usually cheaper too.
Melissa Stansbury
March 19, 2026 AT 03:24I just got my levothyroxine refill and it looked exactly like my old brand - but the bottle said 'authorized generic' and cost $12 instead of $48. I called my pharmacist and she laughed and said, 'You've been paying extra for a sticker.' I feel like I've been lied to my whole life. Why isn't this common knowledge??
cara s
March 19, 2026 AT 05:28It is a fascinating, albeit deeply unsettling, phenomenon that the very entities responsible for the inflated pricing structures of pharmaceuticals - through aggressive patent extensions, evergreening, and marketing monopolies - are now, with bureaucratic precision, deploying authorized generics as a mechanism to maintain market share while ostensibly facilitating cost reduction. The irony is not lost on those of us who have witnessed the commodification of healthcare as a transactional enterprise rather than a public good. One might argue that this is not innovation, but rather a sophisticated form of regulatory arbitrage - a legal loophole dressed in the clothing of consumer benefit. The FDA’s tacit endorsement of this practice, while technically sound, raises profound ethical questions regarding the alignment of corporate incentives with patient welfare. We are not merely consumers; we are vulnerable populations dependent on pharmacological consistency. And yet, the system allows the original manufacturer to retain control over supply, pricing, and perception - all while the public is led to believe they are 'saving money.'
Amadi Kenneth
March 20, 2026 AT 08:54Shameer Ahammad
March 21, 2026 AT 05:01It is imperative to clarify that the practice of authorized generics, while legally sanctioned, constitutes a form of regulatory arbitrage that fundamentally undermines the principles of market competition. The Hatch-Waxman Act was designed to foster genuine generic entry, not to permit brand manufacturers to preemptively suppress competition by deploying their own products under alternative labeling. The FTC's findings, while statistically valid, fail to account for the long-term suppression of market diversity. When the authorized generic exits the market - as it inevitably does - there is no second-tier competitor to maintain price discipline. This is not a consumer-friendly innovation; it is a calculated strategy to delay market liberalization. One must question the integrity of a system that permits such maneuvering under the guise of therapeutic equivalence.
Alexander Pitt
March 22, 2026 AT 17:57Authorized generics are the best option for drugs like levothyroxine, warfarin, or seizure meds. No guesswork. No variable fillers. Same pill, same factory, same quality. If your pharmacist doesn't mention it, ask. You're probably paying more than you need to.
Manish Singh
March 23, 2026 AT 13:35As someone from India where generics are the backbone of healthcare access, I find this whole authorized generic model fascinating - and a little ironic. In the U.S., people are confused because the pill looks identical. Here, we're just happy to get any affordable version. But I see the point: when you're on a drug that's sensitive to tiny changes, consistency matters. I've seen patients switch generics and have seizures. So yeah - if the brand makes the same pill under a cheaper label? That's smart. Not shady. Smart. Just wish more people knew to ask for it.
Nilesh Khedekar
March 23, 2026 AT 19:18okay so i just read this and i'm like... wait so big pharma just made a fake generic? like they're the ones who made the brand and now they're selling it as a generic? that's not a generic that's a trick. and why does the fda even allow this? it's like they're letting the fox guard the henhouse. and now people are confused because their pill looks the same? of course it looks the same - it's the same pill. but they're calling it 'authorized' like that makes it legit. it's not legit - it's just a way to keep profits while pretending to help. i'm not mad. i'm just disappointed. we got scammed again.