ACE Inhibitor & ARB Interaction Checker
This tool checks the safety of combining ACE inhibitors and ARBs based on current medical guidelines and major clinical studies. According to the FDA and major guidelines, combining these drugs significantly increases risks of hyperkalemia and kidney injury.
When it comes to managing high blood pressure, heart failure, or kidney disease in people with diabetes, two classes of drugs are often used: ACE inhibitors and ARBs. They’re both designed to block parts of the same system in your body-the renin-angiotensin system-but they do it in very different ways. That difference matters a lot when it comes to side effects, safety, and what happens when you take them together.
How ACE Inhibitors and ARBs Work
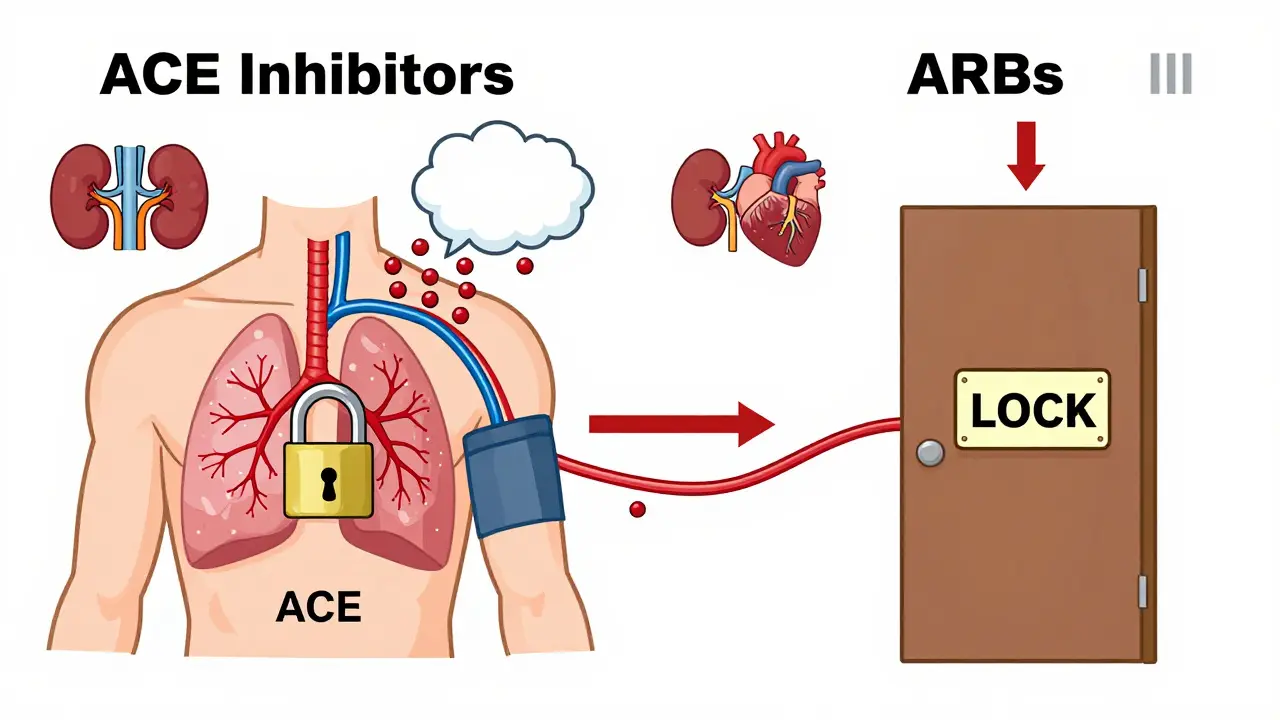
ACE inhibitors like lisinopril, enalapril, and ramipril stop your body from making angiotensin II, a powerful chemical that narrows blood vessels and raises blood pressure. By blocking the enzyme that turns angiotensin I into angiotensin II, these drugs help relax blood vessels and reduce fluid buildup. That’s why they’re so effective for heart failure and diabetic kidney disease.
ARBs-such as losartan, valsartan, and irbesartan-take a different approach. Instead of stopping the production of angiotensin II, they block its action at the receptor level. Think of it like this: ACE inhibitors turn off the faucet, while ARBs lock the door so the water can’t get in, even if it’s still flowing.
This subtle difference has real consequences. ACE inhibitors cause a buildup of bradykinin, a substance that can lead to a persistent dry cough in 10-15% of users. ARBs don’t do that. In fact, only 3-5% of people on ARBs report coughing. That’s why many doctors switch patients from ACE inhibitors to ARBs when the cough becomes unbearable.
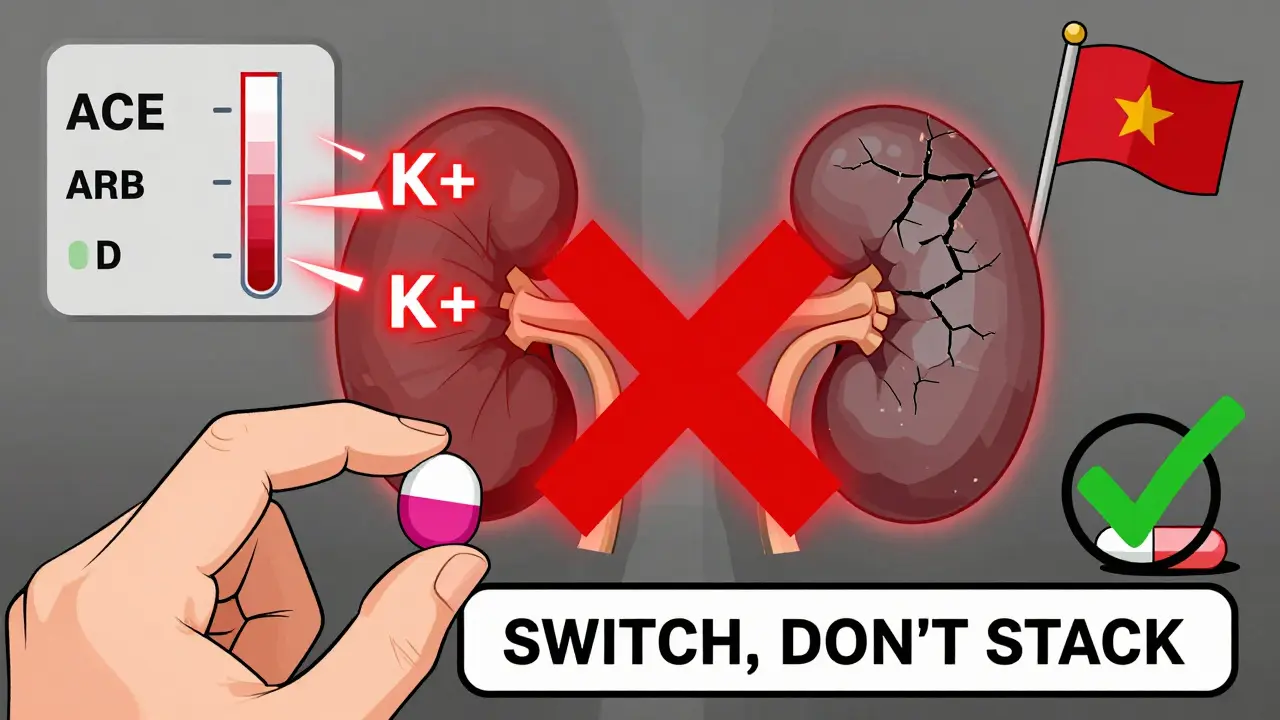
The Big Problem: Combining Them
You might think, if one drug helps, two must help more. But with ACE inhibitors and ARBs, that’s not true. In fact, combining them is risky.
The 2008 ONTARGET trial was a wake-up call. It looked at over 25,000 high-risk patients-mostly with heart disease or diabetes-who were given either ramipril (an ACE inhibitor), telmisartan (an ARB), or both. The combo group saw a small drop in blood pressure and a slight reduction in proteinuria. But here’s the catch: they also had twice the risk of dangerously high potassium levels (hyperkalemia), a 1.8-fold increase in acute kidney injury, and a higher chance of needing dialysis.
The FDA didn’t wait for more data. After studies like ONTARGET and ALTITUDE, they made it clear: don’t combine ACE inhibitors and ARBs. The risks far outweigh any small benefit.
Even in patients with severe kidney disease and heavy proteinuria, studies show that adding an ARB to an ACE inhibitor doesn’t improve survival or prevent kidney failure. Instead, it increases hospitalizations for high potassium, low blood pressure, and kidney shutdown. A 2023 survey of 317 primary care doctors found that 89% had stopped prescribing the combo after the 2018 VA NEPHRON-D trial showed a 27% spike in serious side effects with no improvement in kidney outcomes.
When Is It Ever Okay?
There’s one small gray area. A handful of nephrologists-like Dr. Srinivasan Beddhu from the University of Utah-say that in rare cases, for non-diabetic patients with proteinuria over 1 gram per day who aren’t responding to maximum ACE inhibitor doses, adding an ARB might be considered. But even then, it’s not done lightly.
These patients need weekly blood tests for potassium and kidney function. They’re monitored like hawks. And even in those cases, the benefit is often just a 40-60% drop in proteinuria, not a longer life or less kidney damage.
Most doctors won’t even try it. A Reddit thread from March 2024 showed that 78% of medical residents had seen someone hospitalized because of hyperkalemia from this combo. One nephrologist at Massachusetts General Hospital stopped the combo in 87% of her 215 diabetic kidney patients because potassium rose too high or kidney function dropped too fast.
Side Effects You Can’t Ignore
Both drugs can raise potassium and hurt kidney function, especially in people who are older, have diabetes, or already have reduced kidney function. On average, both classes increase serum potassium by 0.3-0.5 mmol/L. That might sound small, but in someone with borderline kidney disease, it can push them into dangerous territory.
Acute kidney injury happens in 5-10% of high-risk patients within the first few weeks of starting either drug. That’s why doctors check creatinine and potassium levels 1-2 weeks after starting or changing the dose. If creatinine rises more than 30% from baseline, they’ll often hold the drug.
Angioedema-a rare but life-threatening swelling of the face, lips, or throat-occurs in 0.1-0.7% of ACE inhibitor users. With ARBs, it’s rarer: 0.1-0.2%. If you’ve had angioedema on an ACE inhibitor, you’re not a candidate for ARBs. The risk is still there.
Switching Between Them
If you need to switch from an ACE inhibitor to an ARB (or vice versa), don’t just swap them out on the same day. There’s a reason for the 4-week washout period recommended by the ACC. Even though the drugs are different, their effects linger. Taking one right after the other can cause additive drops in blood pressure or sudden spikes in potassium.
But here’s the problem: only 42% of prescribers actually follow that guideline, according to a 2022 JAMA Internal Medicine study. That means a lot of patients are being switched too quickly, putting themselves at risk.
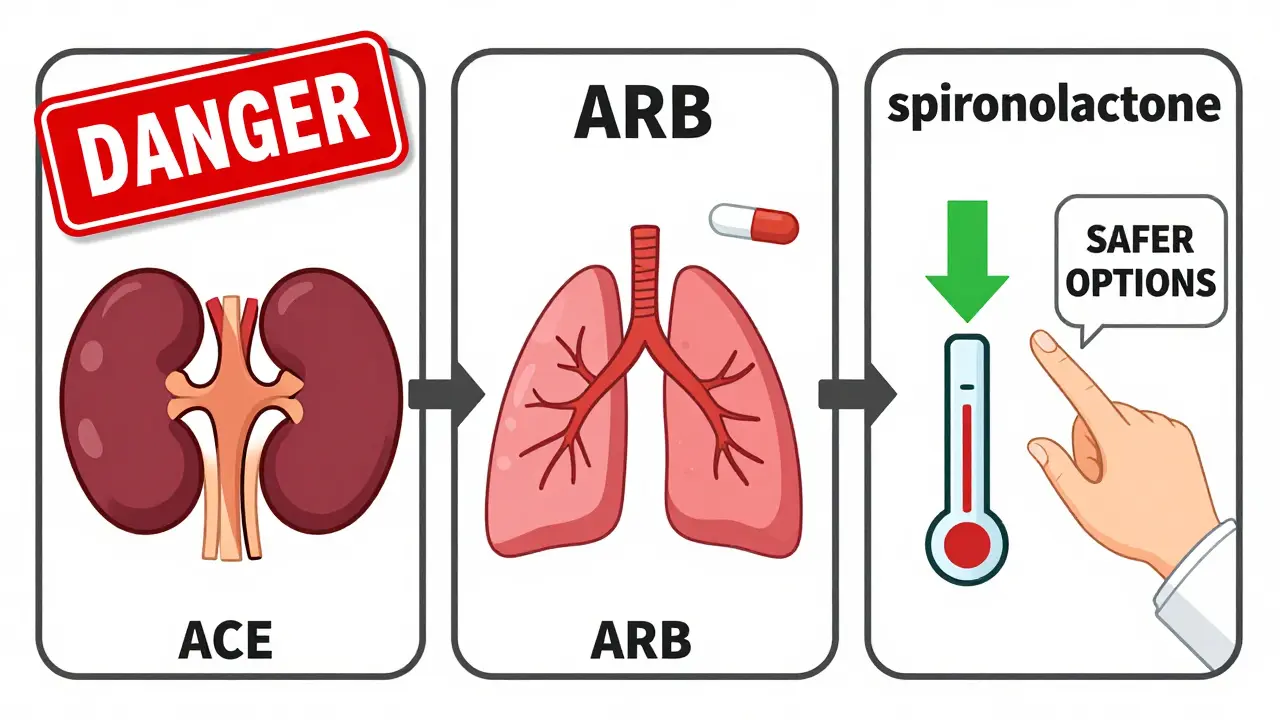
What to Do Instead
If you need stronger blood pressure control or kidney protection beyond what an ACE inhibitor or ARB alone can do, there are safer options.
- For extra kidney protection: Add a low-dose mineralocorticoid receptor antagonist like spironolactone (12.5 mg daily). It reduces proteinuria by 30-40% with fewer risks than an ARB combo.
- For resistant hypertension: Consider a diuretic like chlorthalidone or indapamide. These help flush out extra fluid and potassium.
- For heart failure: Newer drugs like ARNIs (sacubitril/valsartan) have shown better outcomes than ACE inhibitors alone, with fewer side effects.
The 2023 European Society of Cardiology guidelines even suggest switching from an ACE inhibitor to an ARB for patients with persistent high potassium-not adding a second RAS blocker, but replacing the first one. That’s because ARBs are slightly less likely to cause hyperkalemia.
The Bigger Picture
In 2023, ACE inhibitors were prescribed 32.7 million times in the U.S., while ARBs were prescribed 23.6 million times. Lisinopril and losartan are the top two in their classes. But the trend is shifting. More patients are starting on ARBs because they’re better tolerated. And with the FDA’s past recalls of ARBs due to nitrosamine impurities (now mostly resolved), trust in these drugs has recovered.
Looking ahead, the FINE-REWIND trial (2024-2028) is testing whether a very low-dose combo of ACE inhibitor and ARB might be safe for diabetic kidney disease. But even if it works, it’ll be a tiny fraction of patients-and only under strict monitoring.
For now, the message is clear: don’t combine them. If one isn’t working, switch. If you need more, add something else. The risks of stacking these drugs are too high, and the benefits are too small to justify it.
Can I take an ACE inhibitor and ARB together for better blood pressure control?
No. Combining an ACE inhibitor with an ARB increases the risk of serious side effects like hyperkalemia, acute kidney injury, and even dialysis-requiring kidney failure without improving survival or reducing heart attacks or strokes. Major guidelines from the American Heart Association, ACC, and KDIGO all recommend against this combination. The extra 3-5 mmHg drop in blood pressure isn’t worth the risk.
Why do ACE inhibitors cause a dry cough but ARBs don’t?
ACE inhibitors block the enzyme that breaks down bradykinin, a substance that can irritate the airways and cause a persistent, dry cough. ARBs don’t affect bradykinin levels-they only block angiotensin II receptors-so they rarely cause this side effect. That’s why doctors often switch patients from ACE inhibitors to ARBs when the cough becomes disruptive.
What should I do if I’m on both an ACE inhibitor and ARB?
Contact your doctor right away. If you’re on both, you’re at higher risk for dangerous potassium spikes or kidney damage. Your doctor will likely stop one of them-usually the ARB-and monitor your potassium and kidney function closely. Never stop either drug on your own; sudden withdrawal can cause rebound high blood pressure or worsening heart failure.
Are ARBs safer than ACE inhibitors overall?
For most people, yes. ARBs have fewer side effects, especially when it comes to cough and angioedema. They’re also just as effective at lowering blood pressure and protecting the kidneys. But ACE inhibitors have stronger evidence for reducing death in heart failure. So the choice depends on your condition: if you have heart failure, ACE inhibitors may be preferred; if you can’t tolerate the cough, ARBs are a better option.
How often should my potassium and kidney function be checked?
When you start or change the dose of an ACE inhibitor or ARB, get blood tests within 1-2 weeks. If everything’s stable, check every 3 months. If you have diabetes, kidney disease, or are over 65, your doctor may want checks every 1-2 months. High potassium or rising creatinine means the drug may need to be adjusted or stopped.
What are the alternatives if I can’t take ACE inhibitors or ARBs?
Other blood pressure medications include calcium channel blockers (like amlodipine), thiazide diuretics (like hydrochlorothiazide), or beta-blockers (like metoprolol). For kidney protection, mineralocorticoid receptor antagonists (like spironolactone) are a good option. In heart failure, ARNIs (sacubitril/valsartan) have replaced ACE inhibitors in many cases because they’re more effective and better tolerated.

Jacob Hessler
March 24, 2026 AT 11:35And don't get me started on the FDA. Who even are they? Some lab coat zombies telling us what to do. I say keep both. My blood pressure's perfect.
Amber Gray
March 26, 2026 AT 01:46Danielle Arnold
March 27, 2026 AT 22:18My grandma took both. She lived to 94. The real risk is trusting studies written by people who hate fun.
James Moreau
March 29, 2026 AT 13:13Monitoring is key. And sometimes, the 1% who benefit are the ones who get lost in the 99% stats.
J. Murphy
March 29, 2026 AT 18:53Jesse Hall
March 31, 2026 AT 02:29My doc switched me from lisinopril to losartan and my numbers improved. You're not alone. Keep asking questions. ❤️
Donna Fogelsong
April 1, 2026 AT 13:37Also, the ONTARGET trial? Funded by a pharma subsidiary. Read the fine print. They're gaslighting you into dependence.
Sean Bechtelheimer
April 2, 2026 AT 00:14Think about it. Why do you float when you're relaxed? Coincidence? I think not. 🌀
Seth Eugenne
April 2, 2026 AT 16:43You're worth more than a statistic. Your health matters. I'm here if you need to talk. 🤝
Caroline Dennis
April 3, 2026 AT 08:22What we call 'risk' is often just unmeasured resilience. The body doesn't obey guidelines. It obeys biology.
Raphael Schwartz
April 3, 2026 AT 09:01winnipeg whitegloves
April 3, 2026 AT 09:20Marissa Staples
April 3, 2026 AT 12:33Maybe the drugs are just a bandage on a broken bone.
Rachele Tycksen
April 4, 2026 AT 07:29Grace Kusta Nasralla
April 5, 2026 AT 04:13These pills? They're just noise. The real answer is listening. To your breath. To your pulse. To the quiet.